
Published: December 2, 2024 • Reviewed February 6, 2026
Knowing what NOT to do in the days and weeks following a car accident is just as important as knowing what to do. Without realizing it, the things you say or do can easily jeopardize the successful outcome of a personal injury claim. What do you need? Who do you talk to? Or, rather, what do you NOT do after a car accident? Here are some mistakes to avoid after a car accident.

We are pleased to announce that attorneys Brian Franciskato, Dean Nash, and Randy James, have been selected for the 2024 Super Lawyers list.
This is an exclusive list, recognizing no more than five percent of attorneys in the state.

Car accidents are a time of extreme emotion and oftentimes chaotic activity. You are talking with emergency responders and police, witnesses and other occupants of your vehicle, and more. It can take time for both your body and your mind to process what has happened, which may mean that some injuries may be delayed. Two common car accident injuries often delayed are soft tissue and concussions.

According to the 2024 APPA National Pet Owners Survey, 82 million U.S. households own a pet. Of those, 58 million come home to the wagging tail of a dog. In fact, 97% of pet owners consider their pets to be a part of the family. Unfortunately, dog bites and dog attacks do occur even in dog-friendly homes so it’s important to know what to do if you are bitten by a dog.
Quick Dog Bite Facts

Originally Published: August 10, 2016 • Updated/Republished: September 16, 2024
A concussion is a type of brain injury that changes the way the brain normally works and is typically caused by some trauma, either directly or indirectly, to the brain. It is estimated that 1.7 to 3.8 million athletes annually suffer concussions, and at least 5.3 million Americans live with a TBI-related disability. That is one in every 60 people.

The 2024 Missouri Lawyers Media Unsung Heroes have been chosen and we are thrilled to say Amanda Cryderman was selected for the Paralegal category.

Published: September 3, 2024 • Updated: June 9, 2025
Every day, we share the road with large trucks, and it is not uncommon for drivers of smaller vehicles to get a little anxious when a big rig appears in their rearview mirror or alongside them. If you get a little tense sharing the road with large trucks, these safety tips may help you destress and reduce the risk of an accident.

Published: September 16, 2016 • Updated: August 27, 2024
Did you know that one in four people age 65 years or older falls each year? Often, these falls result in hip fractures, broken bones, and head injuries. Fall-related injuries can also be the start of a more serious health issue that can carry a heavy quality-of-life impact, such as long-term disabilities that reduce one’s independence. They are also the leading cause of both fatal and nonfatal injuries for this age group.

The Kansas City Paralegal Association recently announced the results of its Board of Directors elections, and we are proud to say that Amanda Cryderman has been elected as the new Policy Director for the 2024-2026 term!
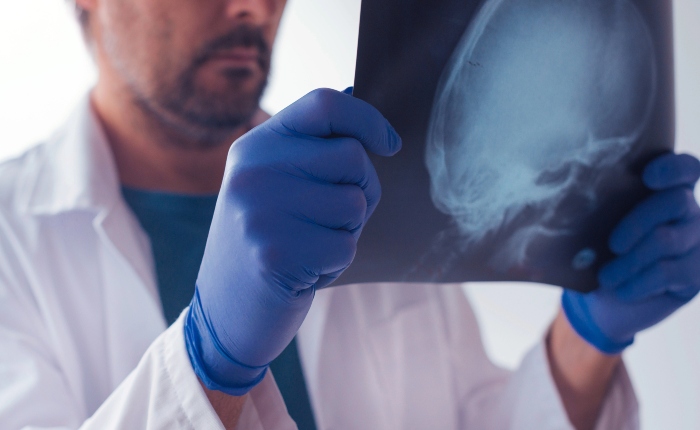
Published: September 16, 2015 • Republished: August 22, 2024 • Updated: March 2, 2026
Every day, 190 people lose their lives to injuries involving a traumatic brain injury (TBI). Among adults 75 and older, TBIs are a leading cause of death and disability, accounting for 28% of all TBI‑related fatalities. Today, an estimated 5.3 million Americans are living with long‑term disabilities caused by a TBI.