
Philips Respironics has now expanded its recall of breathing devices to include certain Trilogy Evo ventilators. In summary,

After a serious accident, you could suffer from a number of physical and/or mental injuries that need medical care, rehabilitation or other forms of care, not just in the short-term, but long-term as well.

Paraquat dichloride, commonly known as “paraquat”, is a popular, fast-acting, non-selective herbicide agent used in the United States. Paraquat is known to be a toxic weed killer that is lethal at low doses, and exposure has been linked to adverse health effects, such as Parkinson’s disease.
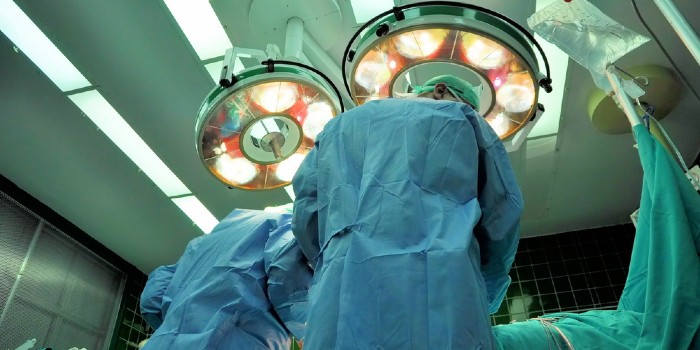
The Exactech hip implant liner (the Connexion GXL) is prone to premature failure, which puts some patients at a higher risk of complications and revision surgery. Exactech’s Connexion GXL hip implant liner was approved for use in primary and revision hip replacement surgeries.
HAVE YOU EXPERIENCED PREMATURE FAILURE OF YOUR DEVICE?

What is a personal injury? This is a legal term that defines cases where one person has been harmed by another. This type of claim is generally based on negligence, where the defendant was careless or had no regard for the safety of others. Common types of personal injury claims include motor vehicle accidents, wrongful death, premises liability. product liability and slip & fall accidents.

Are you still using one of the Philips defective CPAP, BiPAP or other ventilator machines? Have you experienced any serious health problems? Are you aware that the sound abatement foam can break down and lead to health problems?

Gathering the right evidence after a devastating truck accident is critical if you or your passengers are injured. Know that some of the essential evidence that may be needed is different from an accident involving passenger vehicles.

After recently conducting an inspection of a Philips Respironics manufacturing facility, the US FDA released an update related to the June 2021 Class I recall of Philips Respironics CPAP, BiPaP and other ventilators.

We are proud to announce that Brian S. Franciskato and Randy W. James were once again named as Top Attorneys by Thomas Reuters’ Super Lawyers.

In June, a voluntary recall by Philips Respironics, affecting millions of its devices, was issued due to potentially serious health risks related to its CPAP, BiPAP, or ventilator machines’ sound abatement foam. Since then, CPAP lawsuits have been filed by those who have suffered injuries after using the recalled devices.
Contact us for a Free Case Review