
After recently conducting an inspection of a Philips Respironics manufacturing facility, the US FDA released an update related to the June 2021 Class I recall of Philips Respironics CPAP, BiPaP and other ventilators.

In June, a voluntary recall by Philips Respironics, affecting millions of its devices, was issued due to potentially serious health risks related to its CPAP, BiPAP, or ventilator machines’ sound abatement foam. Since then, CPAP lawsuits have been filed by those who have suffered injuries after using the recalled devices.
Contact us for a Free Case Review

In June of this year, Philips Respironics announced a voluntary recall of several models of its CPAP, BiPAP, and other ventilator devices. With 93 proposed class-action lawsuits being filed by the end of September, a request was made to consolidate all claims before one judge for coordinated management as part of a multidistrict litigation. (MDL).
The Judicial Panel of Multidistrict Litigation (JPML) recently heard arguments concerning an MDL. The outcome? Due to the similarity of the claims, the JPML granted motions to consolidate the pretrial proceedings. As of October 8, 2021, there were more than 100 lawsuits filed against Philips.

According to Johns Hopkins, “sleep apnea happens when upper airway muscles relax during sleep and pinch off the airway, which prevents you from getting enough air. Your breathing may pause for 10 seconds or more at a time until your reflexes kick in and you start breathing again.”
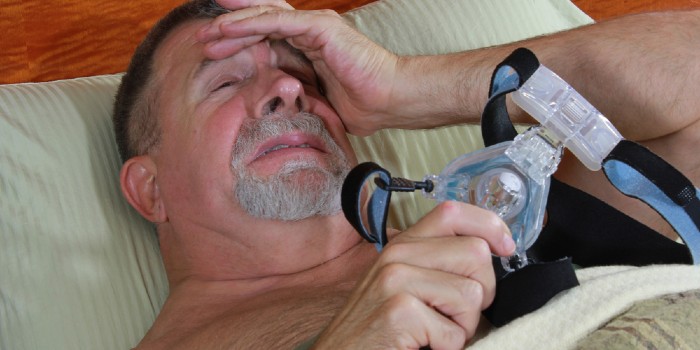
In June of this year, we heard about a voluntary recall made by Philips Respironics of potentially serious health risks related to several of its CPAP, BiPAP and ventilator devices caused by the machines’ sound abatement toxic foam.
START YOUR FREE CASE EVALUATION TODAY

On June 14, 2021, a voluntary recall was announced affecting three to four million Philips Respironics CPAP, BiPAP, and mechanical ventilator devices due to potentially serious health risks related to the machine’s sound abatement foam.
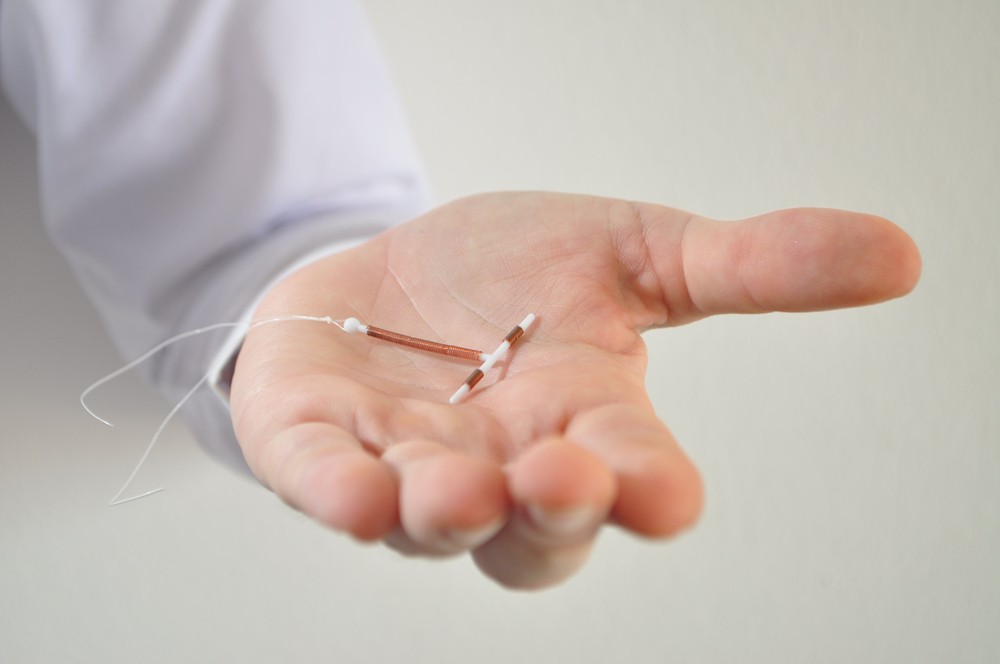
Paragard, a popular intrauterine birth control device used by thousands of women, is prone to breakage and fracturing, which can lead to serious complications. And, there is evidence that the maker, Teva Pharmaceuticals, has been aware of this issue since at least 2013.

One in 15 adults in the United States – roughly 18 million people – suffer from obstructive sleep apnea. One of the most common treatments is using a CPAP machine which keeps the airway open with a gentle, constant stream of pressurized air. However, in June, millions of CPAP and other breathing devices made by Philips Respironics were recalled due to possible health risks and potential dangerous sound abatement foam.

Occupational hearing loss is one of the most common work-related illnesses in the United States. In fact, about 22 million workers are exposed to hazardous noise levels on the job making occupational hearing loss the third most common chronic physical condition among adults. Statistics from NIOSH show:

You may have already heard, Ford is recalling more than 953,000 vehicles worldwide to replace Takata passenger air bag inflators because they can explode and hurl shrapnel.
This recall of Ford vehicles includes over 782,000 vehicles in the United States and is part of the largest series of recalls in U.S. history.